White Paper: Rotary Evaporation
Efficient extraction and distillation require the right temperature control
Rotary evaporators are essential instruments in laboratories for the removal of solvents and the concentration of thermally sensitive compounds. Evaporation takes place at lower temperatures than in conventional distillation processes, as it is performed under vacuum in a rotating flask. This method enables gentle, controlled, energy-efficient, and reproducible distillation.
A key factor in successful rotary evaporation is temperature control technology. It plays a decisive role in ensuring process stability and efficiency. As a leading provider of high-precision temperature control solutions, JULABO supports users in maximizing the performance and precision of rotary evaporators – ideally already during the planning phase, prior to procurement. Selecting the appropriate temperature control solution allows optimization of energy consumption while fully exploiting the system’s application potential.
This white paper outlines the technical fundamentals of rotary evaporation, with particular emphasis on heating and cooling technology. It provides guidance for well-informed decisions when purchasing new equipment or upgrading existing systems and promotes the safe, efficient, and sustainable use of rotary evaporation technology.
A modern rotary evaporator consists of several components that are largely similar across manufacturers and, in combination, enable highly efficient and gentle distillation. The roles played by motion, pressure, and temperature – and the importance of precise temperature control – are explained in this white paper.
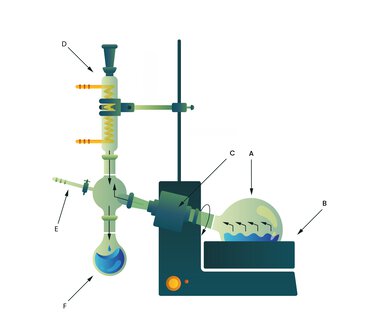
A) Evaporation flask B) Water bath C) Drive motor D) Condenser E) Valve for connection to the vacuum pump F) Receiving flask
1. Development and significance of the rotary evaporator
Modern rotary evaporators are highly precise thermodynamic distillation systems. Temperature, pressure, and rotational speed are electronically controlled and continuously adjusted to the solvent, sample volume, and viscosity. When optimally coordinated, evaporation proceeds efficiently, gently, and reproducibly – representing a significant advancement over conventional distillation methods.
Modern systems enable precise adjustment of evaporation conditions under vacuum. Sensitive natural products, pharmaceutical active ingredients, and biotechnological materials can therefore be concentrated with high yield and minimal thermal stress. Rotary evaporators have consequently become indispensable tools in research, development, and routine analytical laboratories.
The origin of a major innovation
In the early 1940s, the American chemist Lyman C. Craig faced the challenge of isolating compounds from plant extracts, bacterial cultures, and other complex biological mixtures. The separation techniques available at the time proved insufficient.
Craig therefore developed his own method: countercurrent distribution (CCD), also known as Craig distribution. CCD provided high separation efficiency but required large volumes of solvent. As a result, target compounds were obtained in highly diluted form and had to be concentrated by evaporation – a time-consuming step associated with significant thermal input and substance loss. To overcome these limitations and improve the efficiency and gentleness of solvent removal, Craig and his colleagues developed the “Versatile Laboratory Concentration Device” (Craig, Gregory, Hausmann, 1950). This device laid the conceptual foundation for today’s rotary evaporator.
Operating principle of rotary evaporation
A schematic drawing of the original “Versatile Laboratory Concentration Device” illustrates the fundamental operating principle that still applies to modern rotary evaporators:
The solution to be evaporated is placed in a rotating round-bottom flask partially immersed in a heated water bath. Rotation generates a thin, continuous liquid film along the inner wall of the flask. The film thickness is determined by rotational speed and sample viscosity. Reduction of film thickness and expansion of the wetted surface significantly enhance heat transfer between the heating medium, glass wall, and solvent. At the same time, the diffusion path of molecules transitioning from the liquid to the vapor phase is shortened, increasing evaporation rate and effectively preventing local overheating.
Application of vacuum lowers the system pressure and thereby reduces the boiling point of the solvent according to its vapor pressure curve. Evaporation can thus proceed at substantially lower temperatures, minimizing thermal stress and protecting thermolabile compounds. The resulting vapor is transported by the pressure gradient to the condenser, where it condenses on a cooled heat exchanger and collects as liquid distillate in the receiving flask.
Rotary evaporation is therefore a coupled heat and mass transfer process in which rotation, temperature control, pressure regulation, and cooling capacity interact within a closed-loop system.
Temperature, pressure and evaporation
Let us consider, by way of example, the vapor pressure curves of various solvents:
Water boils at 100 °C under atmospheric pressure (1013 mbar). When pressure is reduced to 72 mbar (typical conditions: heating bath 60 °C, condenser 20 °C), the boiling point decreases to approximately 40 °C. Ethanol boils at approximately 78 °C under standard conditions. At a reduced pressure of approximately 175 mbar, evaporation also occurs at 40 °C.
Lowering the boiling point is the defining effect of rotary evaporation. Thermal stress on the sample is reduced, energy consumption decreases, and process speed increases. The condensed vapor liquefies in the condenser, flows downward under gravity, and accumulates in the receiving flask. If condensation is complete, the solvent can be reused.
Rotary evaporation in historical perspective
The fundamental principle developed by Craig and implemented in the first commercial rotary evaporator introduced in 1957 by Büchi and the Basel chemical industry has remained essentially unchanged. What has evolved significantly, however, is the technical implementation. Although rotary evaporation may appear straightforward from the outside, it is in fact a complex thermodynamic process whose performance and quality depend on the precise coordination of all relevant parameters.
Modern laboratory requirements – energy efficiency, process safety, reproducibility, and sustainability – have substantially increased expectations regarding system integration and control technology.
Empirical operation has largely been replaced by sensors, digital control systems, and validated process management. Today, rotation, pressure, and temperature are continuously monitored, synchronized, and controlled within the specific laboratory context.
From this perspective, a rotary evaporator is no longer an isolated device but an integrated system within the laboratory environment. The distillation process represents a closed energy cycle: the heating bath supplies the required energy, while the cooling system removes it in a controlled manner. Only when heating and cooling capacities are precisely matched does evaporation proceed efficiently. Temperature control with accuracy in the tenth-of-a-degree range is therefore not merely desirable – it is a prerequisite for process stability, energy efficiency, and product safety.
2. Heating and cooling – the thermal cycle
Rotary evaporation is a dynamic, closed process. Heat and cooling flows, pressure, and motion continuously interact. The performance of a rotary evaporator cannot be enhanced by rotation alone. Decisive is the precise control of temperature within the overall system.
The heating bath – controlled energy input
Thermal energy is introduced into the system via the heating bath through convective heat transfer. In most applications, water serves as the temperature control medium. For higher temperature requirements, silicone oils are used. Regardless of the medium, the stability of temperature control significantly determines process quality. Even minor deviations can disturb the thermodynamic equilibrium and destabilize evaporation.
Vapor – the energy transport medium
The energy required to transfer the solvent from the liquid to the vapor phase (enthalpy of vaporization) is stored in the vapor and removed from the system during condensation. A stable vacuum ensures uniform heat and mass transfer toward the condenser.
The condenser – controlled energy removal
Within the condenser, the thermal energy contained in the vapor is transferred to a cooling medium, causing the solvent to liquefy. The efficiency of this phase transition depends primarily on the temperature difference between vapor and cooling medium. Only within an optimal temperature window does condensation proceed completely and energy-efficiently.
The closed control loop – prerequisite for efficiency and stability
Heating bath and condenser represent the two thermal poles of the rotary evaporator’s closed-loop system. Temperature, pressure, and flow must be continuously monitored and coordinated to ensure that energy input and removal remain balanced. While the heating bath initiates evaporation, cooling capacity determines whether the process remains stable, reproducible, and sustainable. The cooling system is therefore not merely a passive heat sink but an active control component within the thermal cycle of rotary evaporation. The following chapter examines the resulting requirements for modern cooling solutions and explains why external recirculating coolers represent the current state of the art.
3. The recirculating cooler – precise, efficient and sustainable
The formerly common practice of cooling with tap water is now considered outdated. Fluctuating water temperatures, lack of controllability, and high resource consumption result in unstable processes and conflict with modern requirements for efficiency and sustainability. For this reason, recirculating coolers represent the current state of the art in rotary evaporation.
As a central component of modern rotary evaporator systems, the recirculating cooler ensures precise temperature control of the condenser and stabilizes the entire thermal cycle. It operates actively within a closed system, electronically regulates temperature, and provides constant cooling performance – even under varying ambient conditions or fluctuating vapor loads. This creates reproducible process conditions while simultaneously reducing water and energy consumption.
The refrigeration circuit of a recirculating chiller is based on the principle of a refrigeration system that absorbs heat from a process medium (water, water–glycol mixture, or oil) and dissipates it to the surroundings. It is a closed-loop system that enables a constant temperature of the external device to be cooled (e.g., laboratory equipment, lasers). The cooled medium is pumped through the condenser of the rotary evaporator, where it absorbs the energy released during condensation, and is subsequently returned to the cooler. The circuit operates in a self-regulating manner and independently of external supply conditions.
The high control accuracy of modern recirculating coolers ensures complete and uniform condensation of solvent vapors. In this way, sensitive substances are protected, vapor losses are prevented, the vacuum pump is relieved, and process safety is ensured. The performance of a rotary evaporator can only be guaranteed if the cooling capacity and pump characteristics of the recirculating cooler are matched to the specific application.
Accordingly, the selection of a suitable recirculating cooler comes into focus. Decisive factors are not limited to the selectable temperature range and nominal cooling capacity, but also include control dynamics, delivery pressure, flow rate, and energy efficiency. How these requirements are translated into practical system solutions and which criteria are relevant for selection are explained in the following section using specific equipment concepts.
| Aspect | Recirculating cooler | Conventional cooler (e.g. tap water |
|---|---|---|
| Cooling type | Active, closed cooling system with compressor | Passive flow-through system without temperature control |
| Temperature control | Precisely adjustable, constant (-20 to +85 °C) | Depending on water temperature and line pressure |
| Circulation | Closed circulation, no loss of the temperature control medium | Open circuit, continuous consumption |
| Process stability | High reproducibility, uniform condensation | Variable conditions, inconsistent results |
VALEGRO recirculating coolers – sustainable precision for every application
JULABO recirculating coolers are high-performance solutions for a wide range of cooling applications in laboratory and industrial environments. The VALEGRO series represents modern, compact, and resource-efficient recirculating coolers using natural refrigerants. It has been specifically developed for applications requiring high temperature accuracy, energy efficiency, and ease of operation – across all areas of use, from research and laboratory environments to industrial production.
Speed-controlled compressors and fans in units with cooling capacities of 800 watts and above enable energy-efficient and climate-conscious operation while maintaining constant cooling performance. This can reduce energy consumption by up to 75%, lower operating costs, and support sustainable use in everyday laboratory practice.
In daily operation, the units also demonstrate their practical design features, such as an inclined OLED display, an illuminated level indicator, and an easily accessible, service-friendly ventilation grid. Digital interfaces (RS232, USB-C, optional Ethernet) enable integration into automated laboratory environments and support GLP/GMP-compliant process documentation.
The appropriate unit size for a given application is determined primarily by the required cooling capacity. For standard applications with moderate volumes, VALEGRO models are available with cooling capacities ranging from 0.35 to 2.5 kW and a working temperature range of -20 to +40 °C. More demanding applications are covered by VALEGRO models with an additional heating function and an extended working temperature range of -20 to +85 °C.
For larger and more complex tasks, such as high-volume evaporators or extended tubing systems, VALEGRO models with precisely adjustable, increased pump pressure up to 3.5 bar are ideally suited. VALEGRO advanced versions offer maximum flexibility. With an additional heating function and a more powerful, pressure-controlled pump, they meet even the most demanding application requirements.
The JULABO product portfolio provides a suitable solution for every application – from precise laboratory experiments to continuous temperature control in production environments – offering a balanced ratio of cooling capacity, energy consumption, and footprint. JULABO supports users with expert consultation and modular upgrade options to implement application-specific and sustainably designed cooling solutions over the long term.
VALEGRO model overview
VALEGRO recirculating coolers
with cooling capacities of 0.35 - 1.8 kW. The air-cooled VALEGRO recirculating coolers are environmentally friendly all-rounders for a wide range of cooling and heating tasks from -20 to +40 °C. The pump can be set to different pump stages in a user-friendly way.
All models:
VALEGRO 350, VALEGRO 500, VALEGRO 801, VALEGRO 1001, VALEGRO 1201, VALEGRO 1501, VALEGRO 1801
VALEGRO recirculating coolers with extended temperature range
and cooling capacities from 0.8 - 1.8 kW. The VALEGRO recirculating coolers with additional heating function have an extended operating temperature range of -20 to +85 °C. The pump can be set to different pump stages in a user-friendly way.
All models:
VALEGRO 801H, VALEGRO 1001H, VALEGRO 1201H, VALEGRO 1501H, VALEGRO 1801H
VALEGRO recirculating coolers with increased pump capacity
and cooling capacities of 1.2 - 2.5 kW. The more powerful VALEGRO recirculating coolers are suitable for larger and more complex applications. They are characterized by a precisely adjustable, increased pump pressure of up to 3.5 bar.
All models:
VALEGRO 1203, VALEGRO 1503, VALEGRO 1803, VALEGRO 2503
VALEGRO recirculating coolers with extended temperature range and increased pump capacity
and cooling capacities of 1.2 - 2.5 kW. The VALEGRO recirculating coolers in the extended version meet even the most demanding requirements thanks to their additional heating function and more powerful, pressure-controlled pump. They can be used flexibly and offer maximum safety in use.
All models:
VALEGRO 1203H, VALEGRO 1503H, VALEGRO 1803H, VALEGRO 2503H
4. Key parameters of rotary evaporation
As demonstrated, the performance of a rotary evaporator does not depend on a single adjustment parameter but on the coordinated interaction of multiple variables. Pressure, temperature, rotation, cooling capacity, system tightness, and safety collectively determine process efficiency, reproducibility, and sustainability. Isolated optimization of individual parameters is insufficient. Stable and reliable results can only be achieved from a system-level perspective.
Vacuum – stability before maximization
Vacuum is the primary lever for reducing the boiling point. However, the objective is not maximum vacuum, but a stable, controlled operating range adapted to the specific solvent. Excessive vacuum destabilizes the process and promotes bumping (sudden boiling). Insufficient vacuum reduces efficiency and throughput. Precise vacuum control is therefore a fundamental prerequisite for controlled and reproducible evaporation.
Rotation – uniform film instead of turbulence
Rotation increases the evaporation surface area and prevents localized overheating. It causes the liquid to spread along the inner wall of the flask as a thin, stable film driven by centrifugal force. The objective is to maintain a continuous film. The rotational speed to be set – and considered optimal – depends on the viscosity of the sample: low-viscosity solvents require higher rotational speeds, whereas viscous media require lower speeds. Stable film formation is a prerequisite for uniform heat transfer and consistent evaporation.
Temperature – targeted energy input
Temperature provides the energy required for the phase transition. Heating bath temperature and vacuum level must be precisely matched so that the solvent remains at its boiling point under reduced pressure. Excessively high temperatures lead to product loss and inefficient energy usage. Insufficient temperatures result in extended process times. Precise and dynamic temperature control is therefore essential for reproducible results.
Cooling – ensuring complete condensation
Cooling closes the thermal cycle by reliably removing the energy stored in the vapor. Complete condensation requires a defined temperature difference between vapor and cooling medium. Insufficient cooling leads to vapor losses and unstable pressure conditions. Excessive cooling increases energy consumption unnecessarily. Cooling capacity is therefore a central factor influencing both efficiency and sustainability.
System tightness – foundation for process control
Rotary evaporators operate under vacuum and therefore depend on a leak-tight system. Leaks cause pressure fluctuations, unstable evaporation behavior, and increased energy consumption. Regular leak testing and intact seals and ground-glass joints are essential for stable operation.
Safety – an integral part of process control
Rotary evaporators combine vacuum, elevated temperatures, and glass components. To ensure safe process operation, all parameters must be precisely coordinated, protective mechanisms must function reliably, and maintenance intervals must be observed in accordance with specifications. Modern systems support users through automatic shut-off functions, monitoring features, and ergonomic design. Safety is not an optional addition, but an integral component of efficient evaporation.
In other words, the key parameters relevant to rotary evaporation must be considered collectively. Their interaction determines the quality of the result. Those who understand rotary evaporation as a system and maintain control over all adjustment variables and their interplay ensure stable process conditions and achieve high yields while optimizing energy consumption. This reduces costs and supports environmental sustainability.
Rotary Evaporation in Laboratory Terminology
As in many professional fields, laboratory practice has developed its own terminology. While informal expressions are common in daily laboratory communication, technical documentation typically uses standardized phrasing.
For example:
- “Magnetic stirring bar” is sometimes informally referred to as a “stir bar” or simply a “bar.”
- “Liquid–liquid extraction” may colloquially be shortened to “extraction” in context.
In rotary evaporation, typical expressions include:
- “Evaporate to dryness” – complete removal of solvent under reduced pressure.
- “Concentrate in vacuo” – solvent removal under vacuum.
- “Strip off the solvent” – informal term for solvent removal by rotary evaporation.
- “Dry under a stream of nitrogen” – removal of residual solvent using inert gas.
Such expressions reflect practical laboratory communication while remaining technically precise.
- Nobel Prize Organization. Nomination archive: Lyman Creighton Craig. www.nobelprize.org/nomination/archive/show. php?id=20901 (accessed 16.01.2026).
- Craig, L. C.; Hausmann, W.; Ahrens, E. H. Jr.; Harfenist, E. J. (1951). Automatic Countercurrent Distribution Equipment. Analytical Chemistry, 23(9), 1326-1332.
- Gregory, J. D.; Craig, L. C. (1951). The Analytical Specificity of Countercurrent Distribution. Annals of the New York Academy of Sciences, 53(5), 1015-1030. https://doi.org/10.1111/j.1749-6632.1951.tb48879.x
- Kresge N, Simoni RD, Hill RL (2005) Lyman Creighton Craig: Developer of the Counter-current Distribution Method. Journal of Biological Chemistry 280(7). https://doi.org/10.1016/S0021-9258(19)63136-4
- Craig, L. C.; Gregory, J. D.; Hausmann, W. (1950). Versatile Laboratory Concentration Device. Analytical Chemistry, 22(11), 1462-1464.
- Jensen, W. B. (2008). The Origin of the Rotavap. Journal of Chemical Education, 85(11), 1481-1483.
- University of Wollongong. Rotary Evaporation - Laboratory Notes. https://documents.uow.edu.au/content/groups/ public/@web/@sci/@chem/documents/doc/uow093125.pdf (accessed 16.01.2026)
- Wikipedia. Rotary evaporator. https://de.wikipedia.org/wiki/Rotationsverdampfer (accessed 16.01.2026).
- Büchi Labortechnik AG. Technology: rotary evaporation. https://www.buchi.com/de/wissen/technologien/ rotary evaporation (accessed 16.01.2026).
- Büchi Labortechnik AG. 50 years of rotary evaporators. https://web.archive.org/web/20071117061717/http://www. buechigmbh.de/Chronik.8262.0.html (accessed on 16.01.2026).
- Büchi. Circulating cooler. https://www.buchi.com/de/produkte/instrumente/recirculating-chillers (accessed 16.01.2026)
- Julabo. Recirculating cooler. https://www.julabo.com/de/produkte/umlaufkuehler-umwaelzkuehler/valegro-umlaufkuehler (accessed 16.01.2026)
- IUPAC (International Union of Pure and Applied Chemistry): Green Book - Quantities, Units and Symbols in Physical Chemistry, 3rd Ed., 2007, and ISO 80000-5:2019 "Quantities and units - Part 5: Thermodynamics".